Donation after circulatory death
Overview
Donation after Circulatory Death (DCD), previously referred to as donation after cardiac death or non-heartbeating organ donation, refers to the retrieval of organs for the purpose of transplantation from patients whose death is diagnosed and confirmed using cardio-respiratory criteria.
There are two principal types of DCD, controlled and uncontrolled. Uncontrolled DCD refers to organ retrieval after a cardiac arrest that is unexpected and from which the patient cannot or should not be resuscitated.
In contrast, controlled DCD takes place after death which follows the planned withdrawal of life-sustaining treatments that have been considered to be of no overall benefit to a critically ill patient on ICU or in the Emergency Department.
The clinical circumstances in which DCD can occur are described by the Maastricht classification.
The Maastricht classification of Donation after Circulatory Death
| Category | Type | Circumstances | Typical location |
|---|---|---|---|
| 1 | Uncontrolled | Dead on arrival | Emergency Department |
| 2 | Uncontrolled | Unsuccessful resuscitation | Emergency Department |
| 3 | Controlled | Cardiac arrest follows planned withdrawal of life sustaining treatments | Intensive Care Unit |
| 4 | Either | Cardiac arrest in a patient who is brain dead | Intensive Care Unit |
Worldwide, there is considerable variation in the contributions that DCD makes to deceased donation overall. While some countries have no DCD programmes whatsoever, in others such as the UK, Netherlands and Australia the contributions are significant.
Furthermore, whilst some countries focus principally on controlled DCD (e.g UK, Australia) or uncontrolled DCD (e.g. France, Spain), other countries such as the Netherlands support both forms of DCD.
Figure 1. Relative contributions of donation after brain death (DBD) and donation after circulatory death (DCD) to deceased donation in various countries around the world, as measured by donors per million population (pmp) in 2023. Source: Council of Europe – Transplant Newsletter
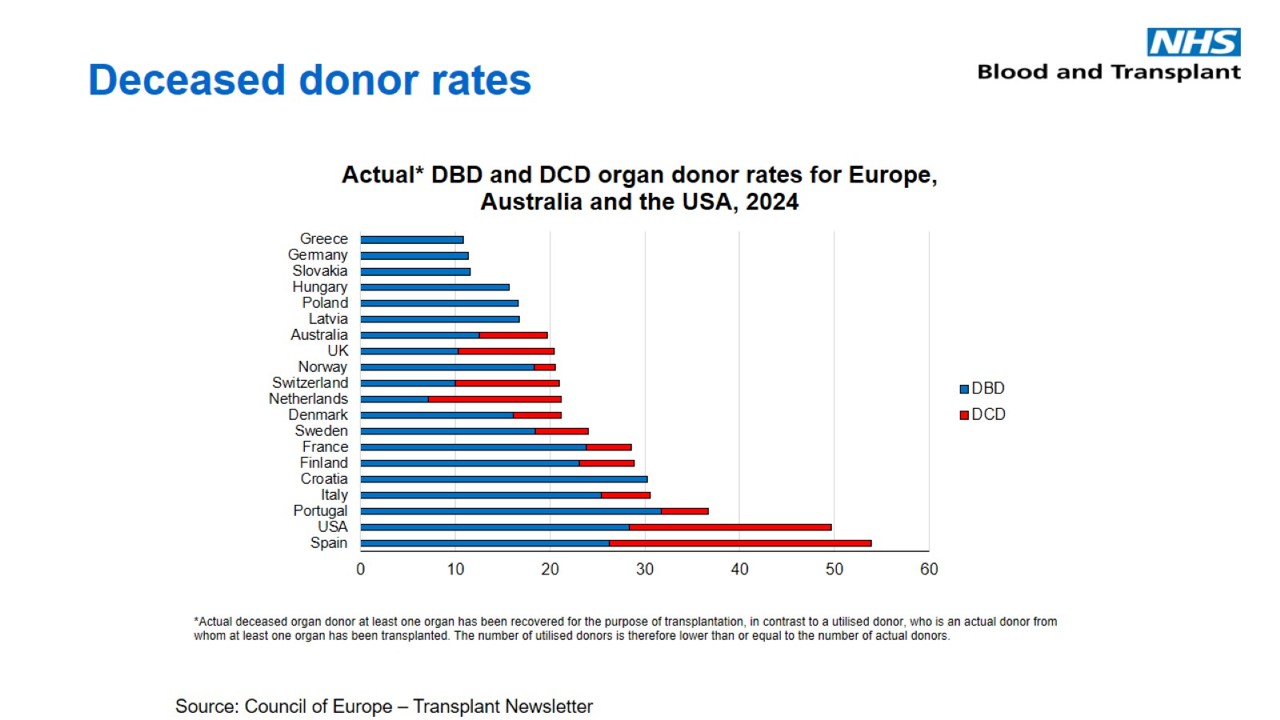
Figure 1. Relative contributions of donation after brain death (DBD) and donation after circulatory death (DCD) to deceased donation in various countries around the world, as measured by donors per million population in 2024. Source: Council of Europe – Transplant Newsletter
The relatively high potential for controlled DCD in the UK is likely to be a reflection of the number of deaths in intensive care that follow a decision to limit or withdraw life-sustaining treatments that are considered to be of no overall benefit to a critically ill patient
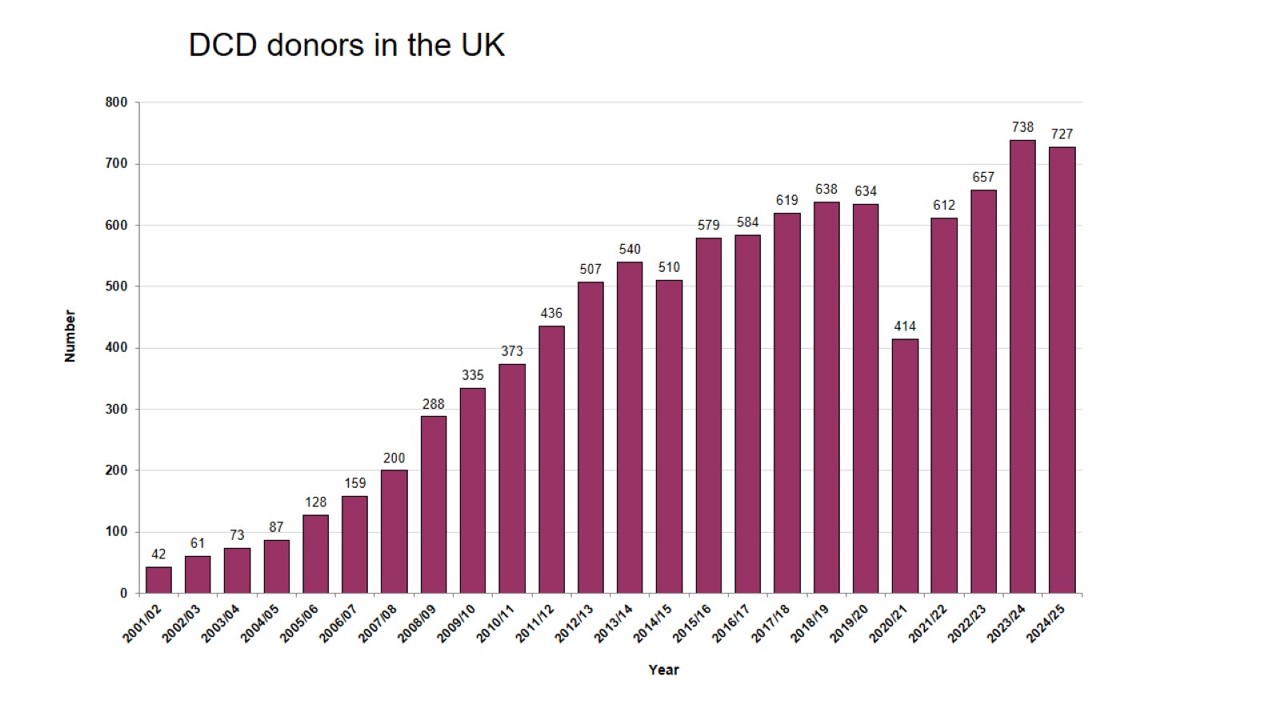
Number of DCD donors in the UK, 2002-03 to 2022-23
DCD donation has increased substantially over the last 20 years, from 87 donors in 2004-2005 to 727 in 2024-2025, representing 52% of all deceased organ donors in this year.
The success of the UK DCD programme can be attributed to the resolution of the apparent legal, ethical and professional obstacles to this model of donation. The underpinning principle of the programme is that donation can on many occasions be legitimately be viewed as part of the care that a person might wish to receive at the end of their lives.
Various publications and professional documents have supported the introduction of controlled DCD programmes into the UK, and they should form the basis for the local policies that describe how this type of donation is incorporated into a patient’s end of life care. Important national documents relating to DCD include:
Professional guidance
This guidance was developed by the British Transplantation Society and Intensive Care Society:
The guidance provides up to date guidance on all aspects of controlled donation after circulatory death, including:
- Decision making
- Care before treatment withdrawal
- Criteria for DCD
- Treatment withdrawal
- Stand down
- Diagnosis of death and post mortem interventions
- Ethical guidance
- Legal guidance
- Legal issues relating to non-heartbeating organ donation, from the Department of Health in England (applicable to practice in England and Wales) (PDF 342KB)
- Guidance on legal issues relevant to donation following cardiac death (applicable to Scotland) (PDF 154KB)
- Professional publications
- Manara AR, Murphy PG, O'Callaghan G. Donation after circulatory death British Journal of Anaesthesia (2012) 108(suppl 1): i108-i121
Contribution of DCD to transplantation in the UK
In the UK, an average of 2.9 transplantable organs are retrieved from DCD donors, compared to 3.6 from DBD donors. The biggest contribution of DCD is to kidney transplantation, with 52% of all deceased donor kidney transplants coming from this source in 2024-2025.
The lower donation potential of DCD donors is in large part a result of the ischaemic injury suffered by solid organs in the time interval between treatment withdrawal and cold perfusion, with the liver and pancreas being particularly vulnerable.
For kidney grafts this is reflected in a higher incidence of delayed graft function (requiring a short period of post-implantation renal support), although long term outcomes are similar to DBD grafts.
However, the consequences of excessive ischaemic injury to the liver are rather more serious, with a higher incidence of graft failure, post-operative morbidity and ischaemic cholangiopathy.
In contrast, DCD donors may be the preferred source of transplantable lungs because the organs have not been exposed to the sympathetic storm suffered by potential DBD donors.
Future developments in controlled DCD are likely to centre around efforts to reduce ischaemic injury and increase the number of lung retrievals from DCD donors, including heart retrieval.
Although some of this is dependent upon post mortem interventions such as regional normothermic recirculation and ex vivo reperfusion, a closer adherence to the standards for the confirmation and diagnosis of death as defined by the Academy of the Medical Royal College report and described in the Consensus statement on DCD from the Intensive Care Society and British Transplantation Society will also make an important contribution by limiting the initial warm ischaemic injury.
For further details on the contribution of DCD to transplantation in the UK, go to NHS Blood and Transplant's latest Transplant Activity report.
DCD heart donation
Due to a combination of factors, the number of hearts available to transplant in the UK falls significantly short of demand. In order to balance the limited supply of donated hearts with the increasing demand, alternative approaches continue to be explored.
In the past it was not possible to facilitate heart donation from donors following circulatory death (DCD), but recent technology developments have enabled this to happen.
The UK is the world leader in DCD heart retrieval and transplantation, to date the UK has carried out over 125 heart transplants. The outcome for recipients are comparable to hearts following DBD transplants and significantly has allowed many more donors to give a lifesaving transplant, an important outcome for donor families.
DCD donors account for a high percentage of the total deceased donor pool in the UK. In an attempt to increase organ supply, a 12-month UK wide pilot has been launched to support cardiothoracic (CT) centres to successfully utilise hearts from DCD donors.
CT teams have worked together to develop and implement the national pilot to facilitate heart retrieval and transplantation from DCD donors. In collaboration with stakeholders, including critical care and abdominal transplant teams, a DCD heart retrieval protocol and passport have been developed to support consistent and successful outcomes in this pilot phase.
Download the UK National Protocol for retrieval of DCD Heart and Lungs
Please note this document may be amended following clinical updates and is intended to be accessed at time of retrieval - most recent versions will be uploaded as updates are made.
